Healthy Skepticism AdWatch
AdWatch illuminates the logical, psychological and pharmacological techniques used in drug advertisements.
October 2003, Australia
Nexium (esomeprazole) from AstraZeneca
Appealing to our desire for power.

Source: Australian Family Physician September 2003: 2 page advertisement inside front cover.
Desire
The Nexium advertisement appeals to our desire for power.
Appeals to desires are common in advertising because they work.[1] These appeals work despite the evidence that we normally do not give them much thoughtful attention. Lack of attention can allow these appeals to sneak into the brain under the radar of critical appraisal.[2]
Unfair comparisons
The Nexium advertisement claims that “Nexium 40mg offers greater healing power that either omeprazole or lansoprazole”. This claim is based on two unfair comparisons.
One of the comparisons is esomeprazole (Nexium) 40mg vs omeprazole (Losec, Acimax, Probitor) 20mg. (The dose of omeprazole is only disclosed in the fine print.)
Omeprazole is a proton pump inhibitor that has been used for years to treat peptic ulcers and gastro-oesophageal reflux disease by reducing gastric acid levels.
Omeprazole 20mg contains about 10mg of S-omeprazole and 10mg of its mirror molecule: R-omeprazole. Both S and R-omeprazole are converted to the same active drug that reduces production of gastric acid by proton pumps. R-omeprazole is absorbed into the body less than S-omeprazole and removed faster, so it may be less effective than S-omeprazole.
Esomeprazole is just a different spelling for S-omeprazole. “Es” = “S-”. AstraZeneca are using a different spelling that makes it less obvious that esomeprazole is just half of their old drug omeprazole (Losec).
It is no surprise that 40mg S-omeprazole in Nexium 40mg is more effective than the 10mg of S-omeprazole plus 10mg of R-omeprazole. However a larger dose of omeprazole would be just as “powerful” as esomeprazole (Nexium) 40mg.
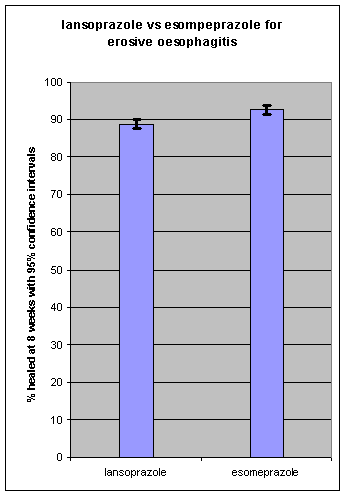
The other comparison is esomeprazole (Nexium) 40mg vs lansoprazole (Zoton) 30mg. (Again the dose of lansoprazole is only disclosed in the fine print.)
The graph below suggests that the difference in the percentage healed at 8 week is too small to be clinically significant.[3] Patients are unlikely to be able to detect a worthwhile difference in relief of symptoms.
Missing comparisons
The advertisement does not compare Nexium with other alternatives. The alternatives that should be considered include other proton pump inhibitors, other drugs and non-drug therapies.
The only trial comparing esomeprazole with pantoprazole that we are aware of was published in September 2003 so it is too recent to be mentioned in the advertisement.[4] That trial suggests that esomeprazole 40mg is unlikely to have any clinically significant advantages over pantoprazole 40mg. This is despite evidence that pantoprazole 40mg is not significantly better than omeprazole 20mg.[5] Both can be true because there is little difference between proton pump inhibitors especially at higher doses. This is because of the law of diminishing returns as shown in the graph below.
We are not aware of any trials comparing esomeprazole with rabeprazole which is the only other proton pump inhibitor currently available in Australia.
Many people with gastro-oesophageal reflux disease can be adequately treated long term with lifestyle changes including elevation of the head of the bed plus antacids or H2 antagonists such as ranitidine (Multiple brands).[6]
Many people with peptic ulcers are better treated with H. pylori bacteria eradication therapy.
General Recommendation
There is no proven clinically significant efficacy or safety advantage of Nexium over fair comparison doses of other proton pump inhibitors.
When a proton pump inhibitor is appropriate, use whichever is the cheapest at the time in the lowest dose that controls the symptoms.
Current Recommendation for Australia
The table below includes all proton pump inhibitors listed in the Pharmaceutical Benefits Scheme Yellow Book effective from 1 August 2003 in order of the cost of a pack of 30 tablets or capsules.
| rabeprazole 10 mg | Pariet | $ 27.60 |
| omeprazole 10 mg | Losec | $ 29.55 |
| pantoprazole 20 mg | Somac | $ 30.43 |
| lansoprazole 15 mg | Zoton | $ 30.98 |
| esomeprazole 20 mg | Nexium | $ 46.19 |
| omeprazole 20 mg | Acimax, Probitor | $ 46.19 |
| rabeprazole 20 mg | Pariet | $ 46.19 |
| omeprazole 20 mg | Losec | $ 47.69 |
| pantoprazole 40 mg | Somac | $ 48.50 |
| lansoprazole 30 mg | Zoton | $ 49.36 |
| esomeprazole 40mg | Nexium | $ 75.26 |
As doses increase prices increase but differences in efficacy diminish. The best value approach may be:
- Commence therapy with lifestyle changes plus esomeprazole 20mg (Nexium). This will quickly control symptoms for most patients.[7]
- If symptoms are not controlled try pantoprazole 40mg (Somac) before using esomeprazole 40mg (Nexium) as a last resort.
- If symptoms are controlled then try lifestyle measures alone or add a H2 antagonist such as ranitidine (Multiple brands) or rabeprazole 10mg (Pariet) for maintenance.
Further reading
Do Single Stereoisomer Drugs Provide Value?
Therapeutics Letter, issue 45, June - September 2002
http://www.ti.ubc.ca/pages/letter45.htm
Jankowski J, Jones R, Delaney B, Dent J.
10-minute consultation: Gastro-oesophageal reflux disease.
BMJ. 2002 Oct 26;325(7370):945.
http://bmj.bmjjournals.com/cgi/content/full/325/7370/945
Feedback
(Feedback will be provided to AstraZeneca and to regulatory agencies. We will take feedback seriously for improving future editions of AdWatch.)
We are longer accepting feedback on this issue of AdWatch since 23 December 2003.
My evaluation of this AdWatch edition is:
No opinion
More helpful than misleading
Neutral
More misleading than helpful
My comments about this AdWatch edition are:
My evaluation of the Nexium advertisement is:
No opinion
More helpful than misleading
Neutral
More misleading than helpful
My comments about the Nexium advertisement are:
Before reading this AdWatch I
Nexium
for gastro-oesophageal reflux disease.
I now plan to
Nexium
.
I
give permission for my feedback to be included in data for research reports.
I am a
The following information is optional.
My name is:
I
want my name sent with my feedback to pharmaceutical companies and regulatory agencies.
Please add my email address, as follows, to the list to be notified about new editions of AdWatch:
(Your email address will not be given to any other organisation. Please check that your email address is correct and notify .(JavaScript must be enabled to view this email address) if your email address changes or if you don’t hear from us within one month.)
Any other information or comments:
1. Scott DK, Ferner RE. ‘The strategy of desire’ and rational prescribing. Brit J Clin Pharmacol 1994;37:217-219.
2. Sutherland M. Advertising and the mind of the consumer. Sydney Allen & Unwin 1993
3. Castell DO, Kahrilas PJ, Richter JE, et al. Esomeprazole (40 mg) compared with lansoprazole (30 mg) in the treatment of erosive esophagitis. Am J Gastroenterol. 2002 Mar; 97(3):575-83.
4. Scholten T, Gatz G, Hole U. Once-daily pantoprazole 40 mg and esomeprazole 40 mg have equivalent overall efficacy in relieving GERD-related symptoms. Aliment Pharmacol Ther. 2003 Sep 15;18(6):587-94.
5. www.health.gov.au/pbs/healthpro/pricing/therelativity.pdf
and
Caro JJ, Salas M, Ward A. Healing and relapse rates in gastroesophageal reflux disease treated with the newer proton-pump inhibitors lansoprazole, rabeprazole, and pantoprazole compared with omeprazole, ranitidine, and placebo: evidence from randomized clinical trials. Clin Ther. 2001 Jul;23(7):998-1017.
6. eTG Complete. Therapeutic Guidelines Ltd. Melbourne July 2003 www.tg.com.au
7. Jankowski J, Jones R, Delaney B, Dent J. 10-minute consultation: Gastro-oesophageal reflux disease. BMJ. 2002 Oct 26;325(7370):945. http://bmj.bmjjournals.com/cgi/content/full/325/7370/945
Indexes:
AdWatch (Australia)
Page views since 15 March 2010: 53644
Comments
Our members can see and make comments on this page.


